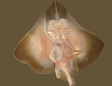
Raja eglanteriaClear-nosed brier skate
Geographic Range
Clearnose skates range along the Atlantic coast of the United States from Massachusetts to southern Florida, as well as in the eastern and northern Gulf of Mexico, from mid-Florida to eastern Texas (22-48°N, 59-91°W). They are rarely found north of Cape Cod, MA. (Luer, et al., 2007; Luna and Binohlan, 2012; Robins and Ray, 1986)
- Biogeographic Regions
- nearctic
- atlantic ocean
Habitat
Clearnose skates live in saltwater bays and estuaries on the coastal sea floor, preferring soft, sandy, near shore substrates. They are typically found in inshore waters less than 100 m deep, but they have been found at depths of up to 330 m. They prefer water temperatures between 5-27°C (most commonly 9-21°C), and salinities of 12-35 ppt. These skates vary their habitat use and depth by season, mainly to remain within their preferred temperature range, but also depending on their activity (breeding versus feeding). ("Sharks, Batoids and Chimaeras of the North Atlantic", 2013; Luna and Binohlan, 2012; McEachran and Dunn, 1998; Piercy, 2010; Schwartz, 1996)
- Habitat Regions
- temperate
- saltwater or marine
- Aquatic Biomes
- benthic
- coastal
- brackish water
- Other Habitat Features
- estuarine
-
- Range depth
- 0 to 330 m
- 0.00 to 1082.68 ft
-
- Average depth
- 20 m
- 65.62 ft
Physical Description
Clearnose skates are a relatively small species of skate, with a maximum recorded length of 84 cm. Together, the head, pectoral fins, and trunk form a broad disc shaped like a flattened spade; the maximum recorded disc width is 48 cm. Individuals in southern parts of the range tend to be smaller. Two dorsal fins are present, separated by a short gap. A single continuous row of stout thorns (33-39 thorns in adults, 14-19 in small juveniles) is present along the midline of the back, extending from the shoulders to the first dorsal fin near the tip of the tail. Smaller thorns are present on the orbital rims and on each shoulder. There are one to three thorns present between the dorsal fins, and at least one row of stout thorns along the lower edges of the tail. The dorsal surface is covered in tooth-like denticles. The outline of the snout is sharply angled, close to 90 degrees. The dorsal surface is brownish to grayish in color and the pectoral fins are marked with dark spots, blotches, and/or bars. A characteristic feature of this species is a translucent space on each side of the snout. The ventral surface is white to cream-colored and free of markings; it is smooth in young individuals and bears a band of spicules along the anterior margins; females may have ventral patches of prickles as well. No geographic or seasonal variation in pigmentation has been reported. These skates have 46-54 teeth in rows set in their upper jaws and 48 teeth in their lower jaws. Males have sharper teeth, set in a different pattern, as well as various dermal spines, to aid in mating. Males are also distinguished by the presence of a pair of claspers (external copulatory organs). ("Sharks, Batoids and Chimaeras of the North Atlantic", 2013; Bigelow and Schroeder, 1953; Fitz and Daiber, 1963; Luna and Binohlan, 2012; Piercy, 2010; Schwartz, 1996)
- Other Physical Features
- ectothermic
- bilateral symmetry
- Sexual Dimorphism
- sexes shaped differently
-
- Range mass
- 3.5 (high) kg
- 7.71 (high) lb
-
- Range length
- 84 (high) cm
- 33.07 (high) in
Development
Development has been studied in great detail for this species. Ova are typically released in pairs, likely one from each ovary. Ova enter a common ostium, then separate again so that each can travel down one of the two oviducts, where they are fertilized. An egg case begins to form before ovulation occurs. Egg cases are, on average, 3.7-4.7 cm wide and 6.4-7.7 cm long. Egg cases have a horn at each of their four corners, each horn with a small groove (respiratory canal) at the base of its lateral edge. Once ova are fertilized, egg case formation is completed. One to thirteen days after fertilization, eggs are deposited. During days one through four after oviposition, a fertilized egg begins cleavage and expansion of the blastodisc. The neural tube and embryonic axis form between days 4 and 7. Pharyngeal pouches and gill filaments develop between days 10 and 28. Respiratory pores in the egg case are unplugged between days 25 and 28, at which time the water is moved through the egg case with continuous tail beats from a developing embryo. During weeks four through seven, fins expand and external filaments transition to internal gills. Epidermal pigmentation develops and yolk absorbtion is completed during weeks 8 through 12. The yolk is absorbed during this time as well. Young hatch around week 12, fully formed and completely independent. (Luer and Gilbert, 1985; Luer, et al., 2007)
Reproduction
In captivity, females have been observed slightly arching their backs, raising their tails, and raising and lowering the posterior margins of both pectoral fins while positioned directly in front of a male. This behavior appears to be an attempt to gain a male's attention, but is not always successful and does not seem to be necessary for the initiation of mating behavior. (Luer and Gilbert, 1985)
A male will approach a female from behind, then bite onto the trailing edge of one of her pectoral fins, holding on with his jaws, as well as with his spines (two sets of dorsal dermal spines, malar spines found lateral to the eye, and alar spines found in rows over the distal portions of each pectoral fin). A male and female have been observed in this position for one to four hours before beginning copulation. To begin copulation, a male then rotates his pelvis underneath a female's tail and pelvic fin, and inserts one clasper into her cloaca. This is a slow process, taking up to an hour to complete. If the male is attached to her right pectoral fin, the right clasper is inserted. If he is attached to her left pectoral fin, he inserts the left clasper. No preference has been documented between initial attachment to the right or left pectoral fin. Once inserted, the clasper expands distally, anchoring it in place. Sperm pass into storage glands, known alternatively as shell glands, oviductal glands, or nidamental glands. Sperm remains viable for up to three months. (Luer and Gilbert, 1985; Luer, et al., 2007)
- Mating System
- polygynandrous (promiscuous)
Clearnose skates are oviparous, and their breeding season is heavily influenced by water temperatures; the optimal water temperature for breeding is 16-22°C. Therefore, different geographical populations may breed at different times. Clearnose skates living near the western coast of Florida breed from December to mid-May. Females mature at 4-6 years of age, while males mature at 2-4 years of age (47-58 cm total length for both sexes). Females store sperm and have been observed producing fertilized eggs up to five weeks following copulation. Sperm are thought to remain viable for up to three months. (Fitz and Daiber, 1963; Ha, et al., 2009; Luer and Gilbert, 1985; Luer, et al., 2007; Luna and Binohlan, 2012; Piercy, 2010)
During oviposition, a female slightly arches her back while contracting the posterior lobes of the pelvic fins ventrally. After shaking the pelvic region from side to side, a single egg is expelled. The second egg is laid several minutes to several hours later. This pelvic shaking often partially buries the egg in the substrate, and sticky material on tendrils at the posterior end of the egg case helps to anchor it. Females lay 23-25 pairs of eggs per season. The total incubation/developmental period for pairs of eggs is about 96 days if the eggs are laid early in the season, but can be as short as 62 days for eggs laid toward the end of the season. Young skates emerge from the egg case at the ruptured anterior end, between the two anterior horns. They are fully formed and independent upon hatching, measuring 13.0-15.0 cm long and 8.4-10.5 cm wide. (Fitz and Daiber, 1963; Luer and Gilbert, 1985; Luer, et al., 2007)
- Key Reproductive Features
- iteroparous
- seasonal breeding
- gonochoric/gonochoristic/dioecious (sexes separate)
- sexual
- fertilization
- oviparous
- sperm-storing
- delayed fertilization
-
- Breeding interval
- Clearnose skates breed once yearly.
-
- Breeding season
- Breeding occurs when when water temperatures are between 16-22°C.
-
- Range number of offspring
- 46 to 70
-
- Average number of offspring
- 60
-
- Range gestation period
- 62 to 96 days
-
- Average gestation period
- 82 days
-
- Average time to independence
- 0 minutes
-
- Range age at sexual or reproductive maturity (female)
- 4 to 6 years
-
- Range age at sexual or reproductive maturity (male)
- 2 to 4 years
Offspring are fully independent upon hatching; no parental care has been documented after oviposition. (Luer and Gilbert, 1985)
- Parental Investment
- no parental involvement
Lifespan/Longevity
Clearnose skates have a lifespan of 5 years or more. (Fitz and Daiber, 1963)
-
- Typical lifespan
Status: wild - 5 (low) years
- Typical lifespan
Behavior
Clearnose skates use a form of benthic locomotion called "true punting", which is somewhat unusual among benthic elasmobranchs. In order to move, they first dig their bilobed pelvic fins into substrate, then retract them caudally, while keeping the body still otherwise. Through this method of locomotion, Clearnose skates are able to perform detailed maneuvers along the substrate, aiding in location and detection of prey while causing little water disturbance, which could alert prey and predators alike. They may also swim in the water column, using a motion that is intermediate between pectoral fin undulation and oscillation. (Macesic and Kajiura, 2010)
-
- Range territory size
- 200 (high) km^2
Home Range
These skates move between spawning and feeding areas, typically within a range of 200 km. Northern populations appear to migrate south and into deeper waters during autumn months, returning during the spring. ("Comprehensive species report: Raja eglanteria", 2012; "Sharks, Batoids and Chimaeras of the North Atlantic", 2013)
Communication and Perception
Clearnose skates have a mechanosensory lateral line system used for detecting water movements. This system is comprised of neuromast receptor organs, composed of hair cells and support cells, which are covered by a gelatinous cupula. Drag from the movement of water displaces the cupula, causing sensory information to be sent to processing centers in the hindbrain. The positioning of the neuromasts helps these animals to detect water movements from currents, predators, prey, or conspecific individuals. They are also electrosensitive, using specialized organs (Ampullae of Lorenzini) to detect electrical signals, while also using electrical discharges socially to communicate. (Maruska, 2001; Sisneros, et al., 1998)
Clearnose skates have low-profile, dorsally positioned eyes. The position of their eyes allows them to bury in the substrate while keeping the eyes uncovered; they are able to see 360° around their dorsal surfaces. Light can also be perceived through their patches of translucent rostral tissue. (McComb and Kajiura, 2008)
Food Habits
Clearnose skates are carnivorous, feeding on decapod crustaceans, bivalves, polychaetes, squids, shrimp, crabs, and fishes. In particular, larger individuals tend to feed preferentially on squid, depending on location and season. ("Sharks, Batoids and Chimaeras of the North Atlantic", 2013; Bigelow and Schroeder, 1953; Piercy, 2010; Sagarese, et al., 2011)
- Primary Diet
-
carnivore
- piscivore
- eats non-insect arthropods
- molluscivore
- Animal Foods
- fish
- mollusks
- aquatic or marine worms
- aquatic crustaceans
Predation
Embryonic clearnose skates both produce (through tail movements related to respiration) and detect (from potential predators) electrical signals. If an embryo senses electrical signals produced by a potential predator, it will halt its tail movements, in order to avoid detection. Once hatched, their elaborate pupillary structures and distinct dorsal markings provide excellent camouflage from predators. Nevertheless, these skates may be prey to other carnivorous fishes, as well as gastropods (as embryos). (Helfman, et al., 2009; Luna and Binohlan, 2012; McComb and Kajiura, 2008; Piercy, 2010; Sisneros, et al., 1998)
- Anti-predator Adaptations
- cryptic
-
- Known Predators
-
- Sand tiger shark (Carcharias taurus)
- Marine snails
Ecosystem Roles
In addition to their ecosystem roles as predators of benthic organisms and prey to larger vertebrates, clearnose skates may serve as hosts to a number of parasites. (Bigelow and Schroeder, 1953; Linton, 1901; Luna and Binohlan, 2012; Piercy, 2010)
- Acanthobothrium paulum (Class Cestoda, Phylum Platyhelminthes)
- Anthobothrium laciniatum (Class Cestoda, Phylum Platyhelminthes)
- Rhinebothrium minimum (Class Cestoda, Phylum Platyhelminthes)
- Rhynchobothrium imparispine (Class Cestoda, Phylum Platyhelminthes)
- Empruthotrema spp. (Class Trematoda, Phylum Platyhelminthes)
- Ascaris rotundata (Class Chromadorea, Phylum Nematoda)
- Echinorhynchus acus (Class Palaeacanthocephala, Phylum Acanthocephala)
Economic Importance for Humans: Positive
Although clearnose skates can be used for food, they are not typically fished and are not considered to be economically important. ("Seafood Handbook - Skate", 2013)
- Positive Impacts
- food
Economic Importance for Humans: Negative
Clearnose skates pose no significant threats to humans, although injury is possible if direct contact with a skate's thorns occurs. (Piercy, 2010)
Conservation Status
This species is currently listed as a species of least concern by the IUCN and has no special status on the U.S. Federal List, CITES, or the State of Michigan list. Clearnose skates remain common despite being frequently caught by fishermen as bycatch. (IUCN, 2013)
-
- IUCN Red List
- Least Concern
-
- US Federal List
- No special status
-
- CITES
- No special status
-
- State of Michigan List
- No special status
Contributors
Molly Miller (author), Indiana University-Purdue University Fort Wayne, Mark Jordan (editor), Indiana University-Purdue University Fort Wayne, Jeremy Wright (editor), University of Michigan-Ann Arbor.
Glossary
- Atlantic Ocean
-
the body of water between Africa, Europe, the southern ocean (above 60 degrees south latitude), and the western hemisphere. It is the second largest ocean in the world after the Pacific Ocean.

- Nearctic
-
living in the Nearctic biogeographic province, the northern part of the New World. This includes Greenland, the Canadian Arctic islands, and all of the North American as far south as the highlands of central Mexico.

- acoustic
-
uses sound to communicate
- benthic
-
Referring to an animal that lives on or near the bottom of a body of water. Also an aquatic biome consisting of the ocean bottom below the pelagic and coastal zones. Bottom habitats in the very deepest oceans (below 9000 m) are sometimes referred to as the abyssal zone. see also oceanic vent.
- bilateral symmetry
-
having body symmetry such that the animal can be divided in one plane into two mirror-image halves. Animals with bilateral symmetry have dorsal and ventral sides, as well as anterior and posterior ends. Synapomorphy of the Bilateria.
- brackish water
-
areas with salty water, usually in coastal marshes and estuaries.
- carnivore
-
an animal that mainly eats meat
- chemical
-
uses smells or other chemicals to communicate
- coastal
-
the nearshore aquatic habitats near a coast, or shoreline.
- cryptic
-
having markings, coloration, shapes, or other features that cause an animal to be camouflaged in its natural environment; being difficult to see or otherwise detect.
- delayed fertilization
-
a substantial delay (longer than the minimum time required for sperm to travel to the egg) takes place between copulation and fertilization, used to describe female sperm storage.
- diurnal
-
- active during the day, 2. lasting for one day.
- ectothermic
-
animals which must use heat acquired from the environment and behavioral adaptations to regulate body temperature
- electric
-
uses electric signals to communicate
- estuarine
-
an area where a freshwater river meets the ocean and tidal influences result in fluctuations in salinity.
- fertilization
-
union of egg and spermatozoan
- food
-
A substance that provides both nutrients and energy to a living thing.
- internal fertilization
-
fertilization takes place within the female's body
- iteroparous
-
offspring are produced in more than one group (litters, clutches, etc.) and across multiple seasons (or other periods hospitable to reproduction). Iteroparous animals must, by definition, survive over multiple seasons (or periodic condition changes).
- migratory
-
makes seasonal movements between breeding and wintering grounds
- molluscivore
-
eats mollusks, members of Phylum Mollusca
- motile
-
having the capacity to move from one place to another.
- natatorial
-
specialized for swimming
- native range
-
the area in which the animal is naturally found, the region in which it is endemic.
- nocturnal
-
active during the night
- oviparous
-
reproduction in which eggs are released by the female; development of offspring occurs outside the mother's body.
- piscivore
-
an animal that mainly eats fish
- polygynandrous
-
the kind of polygamy in which a female pairs with several males, each of which also pairs with several different females.
- saltwater or marine
-
mainly lives in oceans, seas, or other bodies of salt water.
- seasonal breeding
-
breeding is confined to a particular season
- sexual
-
reproduction that includes combining the genetic contribution of two individuals, a male and a female
- solitary
-
lives alone
- sperm-storing
-
mature spermatozoa are stored by females following copulation. Male sperm storage also occurs, as sperm are retained in the male epididymes (in mammals) for a period that can, in some cases, extend over several weeks or more, but here we use the term to refer only to sperm storage by females.
- tactile
-
uses touch to communicate
- temperate
-
that region of the Earth between 23.5 degrees North and 60 degrees North (between the Tropic of Cancer and the Arctic Circle) and between 23.5 degrees South and 60 degrees South (between the Tropic of Capricorn and the Antarctic Circle).
- vibrations
-
movements of a hard surface that are produced by animals as signals to others
- visual
-
uses sight to communicate
References
Department of Commerce and Labor. A biological survey of the waters of Woods Hole and vicinity. Bulletin of the Bureau of Fisheries: Volume 31. Washington, D.C.: United States Government Printing Office. 1911.
2012. "Comprehensive species report: Raja eglanteria" (On-line). NatureServe. Accessed July 10, 2013 at http://www.natureserve.org/explorer/servlet/NatureServe?searchName=Raja+eglanteria.
2013. "Seafood Handbook - Skate" (On-line). Accessed April 04, 2013 at http://www.seafoodsource.com/seafoodhandbook.aspx?id=10737418940.
Food and Agriculture Organization of the United Nations. Sharks, Batoids and Chimaeras of the North Atlantic. FAO Species Catalogue for Fishery Purposes No. 7. Rome, Italy: Food and Agriculture Organization of the United Nations. 2013. Accessed July 09, 2013 at http://www.fao.org/docrep/017/i3178e/i3178e.pdf.
Anderson, W., P. Dasiewicz, S. Liban, C. Ryan, J. Taylor, M. Grosell, D. Weihrauch. 2010. Gastro-intestinal handling of water and solutes in three species of elasmobranch fish, the white-spotted bamboo shark, Chiloscyllium plagiosum, little skate, Leucoraja erinacea and the clear nose skate Raja eglanteria. Comparative Biochemistry and Physiology, Part A, 155: 493-502. Accessed July 08, 2013 at https://www.rsmas.miami.edu/users/grosell/PDFs/2010%20Anderson%20et%20al..pdf.
Bigelow, H., W. Schroeder. 1953. Fishes of the Gulf of Maine. Washington: United States Government Printing Office.
Fitz, E., F. Daiber. 1963. An introduction to the biology of Raja eglanteria Bose 1802 and Raja erinacea Mitchill 1825 as they occur in Delaware Bay. Bulletin of the Bingham Oceanographic Collection, 18/3: 69-97.
Ha, D., C. Luer, J. Sulikowski. 2009. "Raja eglanteria" (On-line). International Union for Conservation of Nature and Natural Resources. Accessed July 08, 2013 at http://www.iucnredlist.org/details/161658/0.
Helfman, G., B. Collette, D. Facey, B. Bowen. 2009. The Diversity of Fishes: Biology, Evolution, and Ecology. West Sussex, UK: John Wiley & Sons.
IUCN, 2013. "The IUCN Red List of Threatened Species. Version 2013.1" (On-line). Accessed July 13, 2013 at http://www.iucnredlist.org.
Linton, E. 1901. Parasites of Fishes of the Woods Hole Region. Washington, D.C.: United States Government Printing Office.
Luer, C., P. Gilbert. 1985. Mating behavior, egg deposition, incubation period, and hatching in the clearnose skate, Raja eglanteria. Environmental Biology of Fishes, 13/3: 161-171. Accessed July 08, 2013 at http://link.springer.com.proxy.lib.umich.edu/content/pdf/10.1007%2FBF00000926.pdf.
Luer, C., C. Walsh, A. Bodine, J. Wyffels. 2007. Normal embryonic development in the clearnose skate, Raja eglanteria, with experimental observations on artificial insemination. Environmental Biology of Fishes, 80/2-3: 239-255. Accessed July 08, 2013 at http://link.springer.com.proxy.lib.umich.edu/content/pdf/10.1007%2Fs10641-007-9219-4.pdf.
Luna, S., C. Binohlan. 2012. "Raja eglanteria Clearnose skate (Bosc, 1800)" (On-line). FishBase. Accessed July 08, 2013 at http://www.fishbase.org/Summary/speciesSummary.php?ID=1252&AT=clearnose+skate.
Macesic, L., S. Kajiura. 2010. Comparative Punting Kinematics and Pelvic Fin Musculature of Benthic Batoids. Journal of Morphology, 271/10: 1219-1228. Accessed July 08, 2013 at http://onlinelibrary.wiley.com.proxy.lib.umich.edu/doi/10.1002/jmor.10865/full.
Maruska, K. 2001. Morphology of the mechanosensory lateral line system in elasmobranch fishes: ecological and behavioral considerations. Environmental Biology of Fishes, 60/1-3: 47-75. Accessed July 08, 2013 at http://link.springer.com.proxy.lib.umich.edu/content/pdf/10.1023%2FA%3A1007647924559.pdf.
McComb, D., S. Kajiura. 2008. Visual fields of four batoid fishes: a comparative study. The Journal of Experimental Biology, 211: 482-490. Accessed July 08, 2013 at http://jeb.biologists.org.proxy.lib.umich.edu/content/211/4/482.full?sid=818cf710-a9db-463d-8e5a-6f7fa7a5efcc.
McEachran, J., K. Dunn. 1998. Phylogenetic analysis of skates, a morphologically conservative clade of elasmobranchs (Chondrichthyes: Rajidae). Copeia, 2: 271-290. Accessed July 08, 2013 at http://www.jstor.org/stable/1447424.
Piercy, A. 2010. "Clearnose Skate" (On-line). Ichthyology at the Florida Museum of Natural History. Accessed April 04, 2013 at http://www.flmnh.ufl.edu/fish/Gallery/Descript/clearnoseskate/clearnoseskate.html.
Robins, C., G. Ray. 1986. A field guide to Atlantic coast fishes of North America. Boston, U.S.A.: Houghton Mifflin Company.
Sagarese, S., R. Cerrato, M. Frisk. 2011. Diet Composition and Feeding Habits of Common Fishes in Long Island Bays, New York. Northeastern Naturalist, 18/3: 291-314. Accessed July 08, 2013 at http://www.bioone.org.proxy.lib.umich.edu/doi/full/10.1656/045.018.0304.
Schwartz, F. 1996. Biology of the Clearnose Skate, Raja eglanteria, from North Carolina. Biological Sciences, 2: 83-95.
Sisneros, J., T. Tricas, C. Luer. 1998. Response properties and biological function of the skate electrosensory system during ontogeny. Journal of Comparative Physiology A, 183/1: 87-99. Accessed July 10, 2013 at http://www.ncbi.nlm.nih.gov/pubmed/9691481.